
Thus, by discarding aneuploid embryos the possibility exists that we could be denying some women the opportunity of having a baby. So clearly, summarily discarding all aneuploid embryos as a matter of routine we are sometimes destroying some embryos that might otherwise have “autocorrected” and gone on to develop into normal offspring. In fact, I am personally aware of several such cases having occurred in my own practice. “autocorrect”) and then go on to develop into chromosomally normal offspring. However, a growing body of evidence suggests that following embryo transfer, some aneuploid embryos will in the process of ongoing development, convert to the euploid state (i.e. those with an irregular quota of chromosomes) be disposed of. Many IVF programs that offer PGS/PGT-A services, require that all participating patients consent to all their aneuploid embryos (i.e. Since then PGS has grown dramatically in popularity such that it is now widely used throughout the world. We subsequently reported on a 2-3-fold improvement in implantation and birth rates as well as a significant reduction in early pregnancy loss, following IVF. those that have a full component of chromosomes) to the uterus. More than 15 years ago, we were the first to introduce full embryo karyotyping (identification of all 46 chromosomes) through preimplantation genetic sampling (PGS) as a method by which to selectively transfer only euploid embryos (i.e. 
Chromosomal abnormalities may arise during germ cell and/or preimplantation embryo development and represents a major cause of early pregnancy loss. 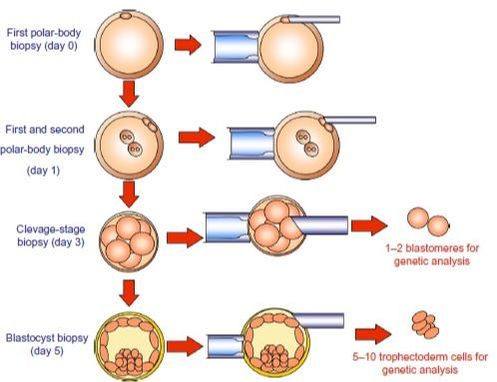
doi: 10.1002/pd.4759.Human embryo development occurs through a process that encompasses reprogramming, sequential cleavage divisions and mitotic chromosome segregation and embryonic genome activation. Detection and quantitation of chromosomal mosaicism in human blastocysts using copy number variation sequencing. Ruttanajit T., Chanchamroen S., Cram D.S., Sawakwongpra K., Suksalak W., Leng X., Fan J., Wang L., Yao Y., Quangkananurug W. Outcomes in pregnancies with a confined placental mosaicism and implications for prenatal screening using cell-free DNA. Grati F.R., Ferreira J., Benn P., Izzi C., Verdi F., Vercellotti E., Dalpiaz C., D’Ajello P., Filippi E., Volpe N., et al. Analysis of implantation and ongoing pregnancy rates following the transfer of mosaic diploid-aneuploid blastocysts. doi: 10.1016/j.fertnstert.2017.05.002.įragouli E., Alfarawati S., Spath K., Babariya D., Tarozzi N., Borini A., Wells D. Detailed investigation into the cytogenetic constitution and pregnancy outcome of replacing mosaic blastocysts detected with the use of high-resolution next-generation sequencing. Munné S., Blazek J., Large M., Martinez-Ortiz P.A., Nisson H., Liu E., Tarozzi N., Borini A., Becker A., Zhang J., et al. Why do euploid embryos miscarry? A case-control study comparing the rate of aneuploidy within presumed euploid embryos that resulted in miscarriage or live birth using next-generation sequencing. Maxwell S.M., Colls P., Hodes-Wertz B., McCulloh D.H., McCaffrey C., Wells D., Munné S., Grifo J.A. Meta-analysis mosaic embryo transfer (MET) multi-center preimplantation genetic testing for aneuploidies (PGT-A) prospective. This is the first multi-center prospective study reporting a full MET pregnancy outcome with complementary information from prenatal genetic testing as compared to euploid and non-PGT cohorts. Longitudinal investigation on one MET pregnancy evidenced the aneuploidy depletion hypothesis. 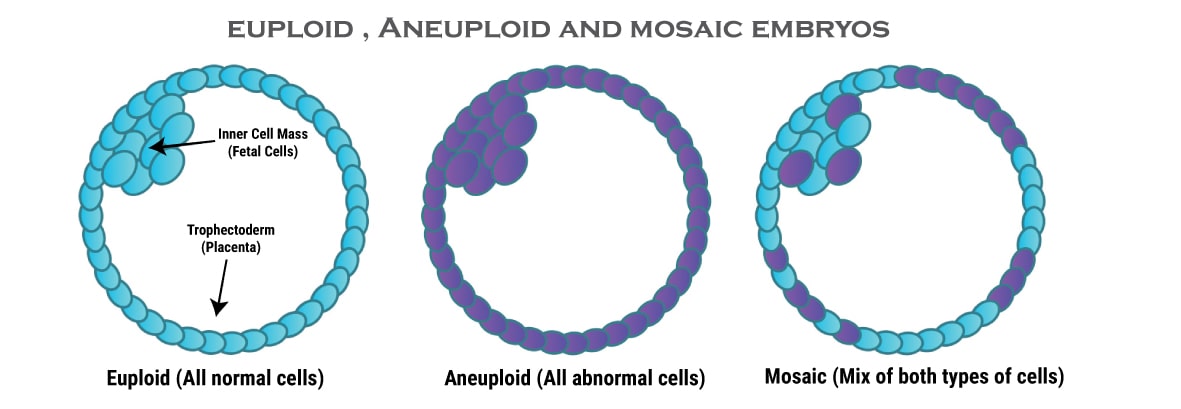
All 37 MET live births were confirmed viable, among which 8 completed prenatal genetic testing with normal results. Our meta-analysis concluded that METs gave rise to pregnancies but were associated with a reduced ongoing/live birth rate and a higher miscarriage rate. Pregnancy losses after METs were different between embryos carrying numerical and segmental chromosomal abnormalities ( p = 0.04). Mosaic embryo transfers (METs) resulted in a significantly lower clinical pregnancy rate (40.1% versus 59.0% versus 48.4%), lower ongoing/live birth rate (27.1% versus 47.0% versus 35.1%) and higher miscarriage rate (33.3% versus 20.5% versus 27.4%) than euploid and non-PGT transfers, respectively. This is a multi-center prospective cohort study on 137 mosaic, 476 euploid and 835 non-preimplantation genetic testing (non-PGT) embryos from three in vitro fertilization (IVF) providers of three countries in Asia, applying the same preimplantation genetic testing for aneuploidies (PGT-A) reporting criteria. Expanded research is warranted to evaluate its clinical significance. Recent studies provided evidence on the viability of mosaic embryos by reporting pregnancy outcomes. Chromosomal mosaicism is at high occurrence in early developmental-stage embryos, but much lower in those at prenatal stage.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
